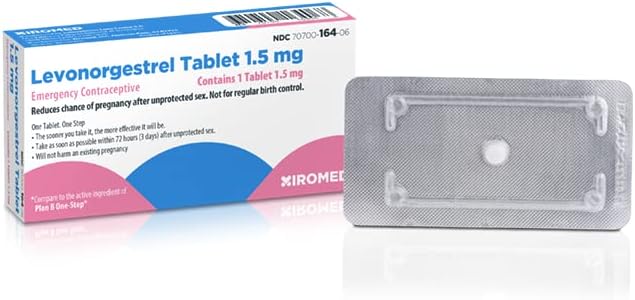
Opill, Daily Birth Control Pills, Over-the-Counter Progestin-Only Oral Contraceptive, FDA-Approved & Estrogen-Free, 84 Count
 Limited Time Sale
Limited Time Sale$21.79 cheaper than the new price!!
Free cash-on-delivery fees for purchases over $99
Product details
| Management number | 211990165 | Release Date | 2026/04/06 | List Price | $14.52 | Model Number | 211990165 | ||
|---|---|---|---|---|---|---|---|---|---|
| Category | |||||||||
Opill is your powerful ally in birth control. Its the first over the counter, full prescription strength, daily oral contraceptive pill that is FDA-approved. Suitable for most* people who can become pregnant, Opill uses one hormone, progestin (norgestrel), and is estrogen-free. Opill is the most effective over the counter birth control** method for women and people who can become pregnant. When used as directed, this daily birth control pill is 98% effective at preventing pregnancy and starts working 48 hours after you take your first pill.*** The best part? You don't need a doctor's appointment or a prescription before you begin using it, so it's a great option for those without health insurance or frequent access to medical services. Get progestin-only daily birth control pills without a prescription. No prescription. No appointment. No hassle. FDA approved. *Do not use Opill if you have or ever had breast cancer. See Opill label for list of warnings. ** When used as directed. ***Be sure to use a barrier method like condoms for the first two days to prevent pregnancy.
- OTC BIRTH CONTROL PILL: Opill is the first over the counter, daily oral contraceptive pill that is available without a prescription. No prescription. No appointment. No hassle.
- PROGESTIN-ONLY: Opill is progestin-only and can be taken by most* people who can get pregnant. *Do not use Opill if you have or ever had breast cancer. See Opill label for list of warnings.
- FDA-APPROVED: Opill is the first prescription-free, daily birth control pill approved by the FDA for over-the-counter use, adding convenience and increasing accessibility for those who need it most.
- FULL PRESCRIPTION STRENGTH: Opill is the most effective over-the-counter contraceptive available for people who can become pregnant when used as directed.
- 98% EFFECTIVE: Our estrogen-free, daily contraceptive pill is 98% effective at preventing pregnancy when taken as directed at the same time every day, providing birth control in your control.
- HSA and FSA eligible product
| UPC | 850054278036 |
|---|---|
| Flavor | Unflavored |
| Item Form | Tablet |
| Brand Name | Opill |
| Model Name | OPILL |
| Unit Count | 84 Count |
| Dosage Form | Tablet |
| Item Weight | 0.13 Pounds |
| Manufacturer | Perrigo |
| Model Number | LPB27803 |
| Container Type | Bottle |
| Number of Items | 1 |
| Target Audience | Women |
| Product Benefits | Birth Control |
| Net Content Count | 84 Capsule |
| Active Ingredients | Norgestrel 0.075 mg |
| Additional Features | Over-The-Counter |
| Special Ingredients | Norgestrel |
| Age Range Description | Adult |
| Specific Uses For Product | To prevent pregnancy. |
| Global Trade Identification Number | 00850054278036 |
Correction of product information
If you notice any omissions or errors in the product information on this page, please use the correction request form below.
Correction Request Form